

Current Research Areas
Gene Therapy
Baculovirus is an insect virus that is harmless to humans but can
efficiently deliver genes (transduce) into a wide variety of mammalian
cells, therefore we are focusing on the development of baculovirus vectors
for gene therapy. We have optimized the baculovirus-mediated gene transfer
protocols as well as developed new titration, production and purification
methods. We employ baculovirus to effectively transduce various cell lines
and stem cells including bone marrow-derived mesenchymal stem cells (BMSCs)
and adipose-derived stem cells (ASCs). We have developed novel FLPo/Frt and
Cre/loxP-based hybrid baculovirus to prolong and enhance
baculovirus-mediated transgene expression. We are also taking advantage of
synthetic biology concepts and CRISPR technologies to develop novel
baculovirus vectors for diverse applications.
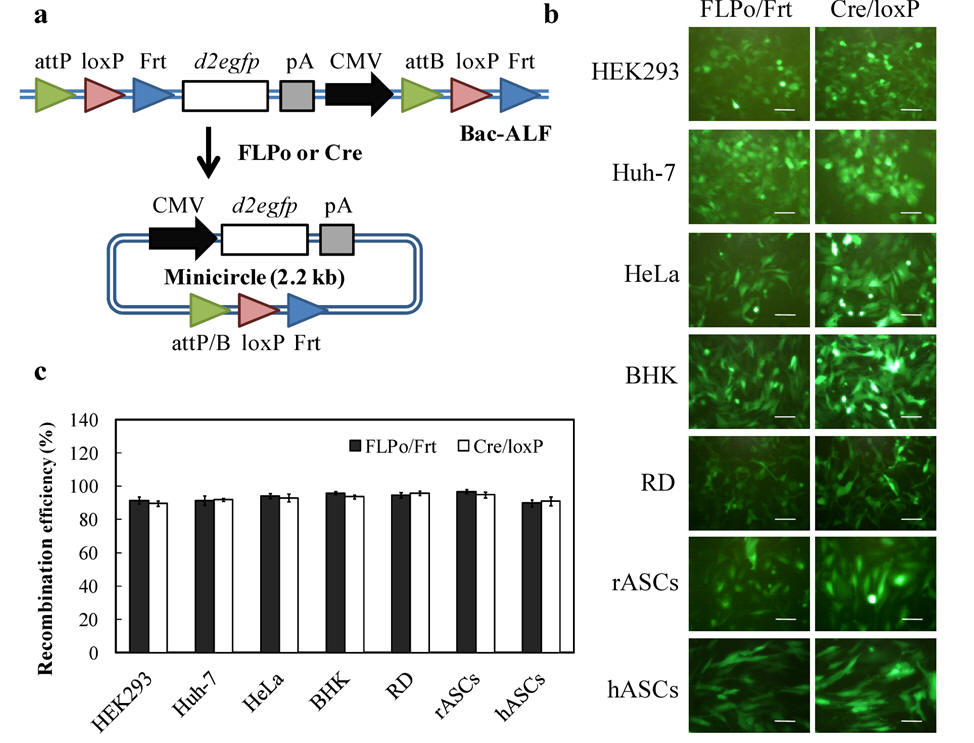
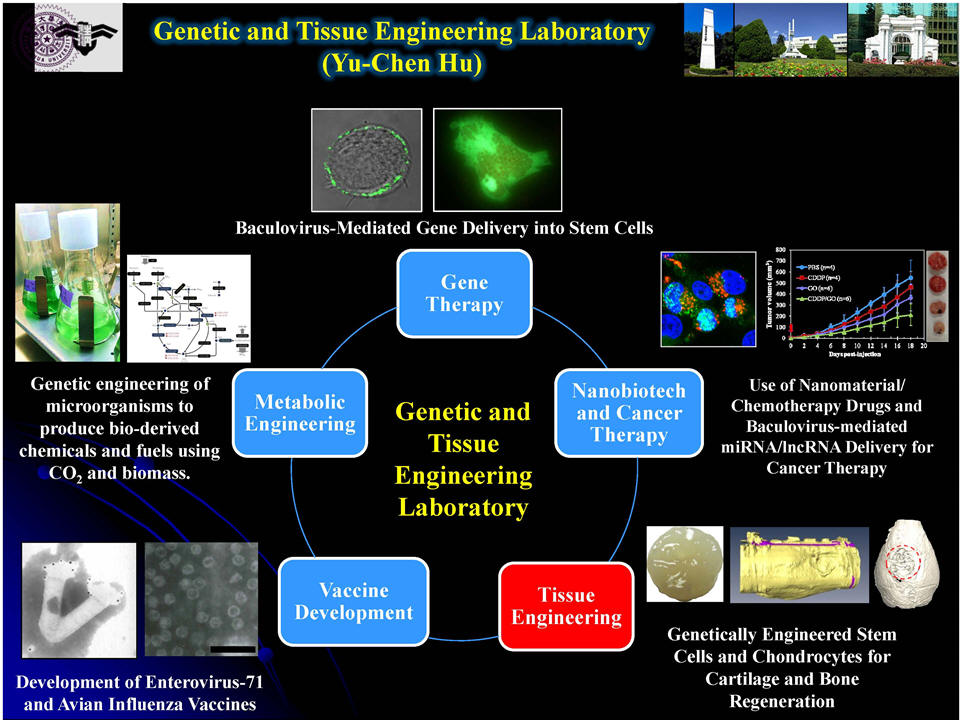
Fig. 1. Development of hybrid
baculoviral vector system for effective gene delivery into various cells and
prolonged/enhanced gene expression. (a)
Schematic illustration of how FLPo (or Cre)-mediated excision of gene
cassette from baculovirus can lead to formation of DNA minicircle.
(b-c)
Such hybrid baculovirus vectors can confer efficient recombination and DNA
minicircle formation, as evidenced by fluorescence microscopy (b) and flow
cytometry (c) analyses.
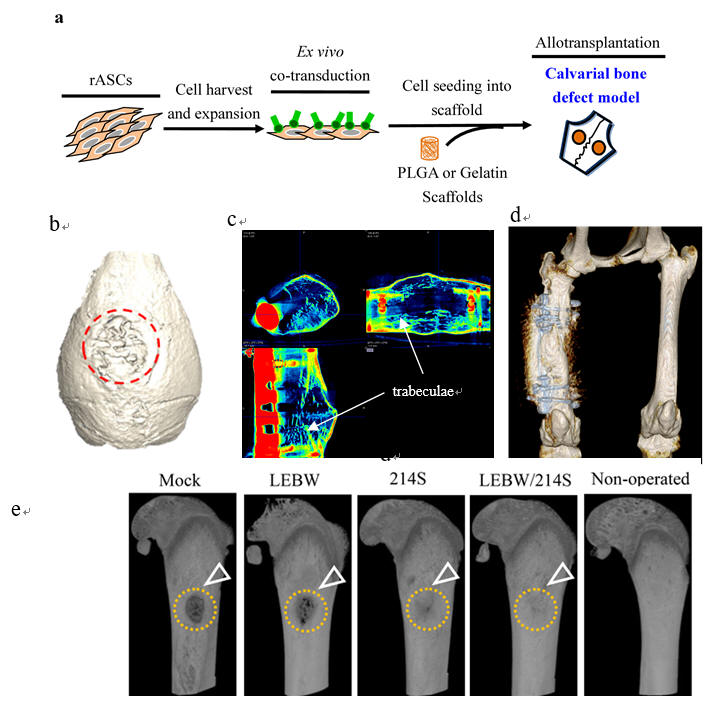
Fig. 2. Baculovirus-engineered adipose-derived stem cells (ASC) repair bone defects at the calvaria and femora. (a) The rabbit ASCs (rASCs) were co-transduced with baculoviruses expressing appropriate transgenes (e.g. BMP-2 and microRNA), seeded to scaffolds and transplanted into defects. (b-e) The baculovirus-engineered cells repair the large defects at the calvaria and femora.
Vaccine
Development
Another important research area in our lab is the development
of vaccines against infectious diseases. One approach we adopt is to produce
recombinant virus-like particles (VLP) of enterovirus 71 (EV71) via genetic
engineering approach. The VLPs are devoid of viral RNA but the structural
proteins on the VLP theoretically can elicit strong immune responses. We
have engineered baculoviruses for infection of insect cells and production
of the recombinant proteins. After purification and injection, our EV71 VLP
induces long-lasting antibody and T cells responses in mice that confer
protection to mice from virus infection, implicating its potential as a
vaccine against EV71. Using the same platform, we have developed vaccines
against porcine circovirus 2 (PCV2) and poliovirus. Another approach we
adopt is to genetically modify baculovirus to display and/or express avian
influenza virus hemagglutinin and inject the baculovirus into animal models,
to induce acquired and innate immunity in vivo. We are also developing new
hybrid baculovirus-based vaccines to potentiate the vaccine efficacy.
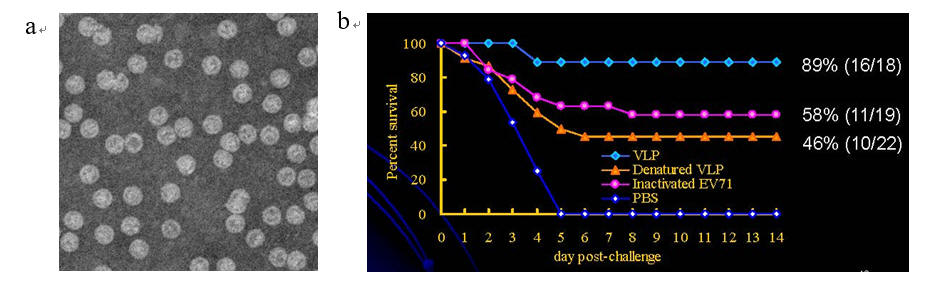
Fig. 3. EV71 VLP produced from baculovirus/insect cell system confers protection against virus infection. (a) The electron micrograph shows the icosahedral morphology of VLP (27 nm in diameter). (b) After immunization, the VLPs confer higher protection (89% survival rate) than other vaccine formats (denatured VLP and inactivated EV71) after virus challenge.
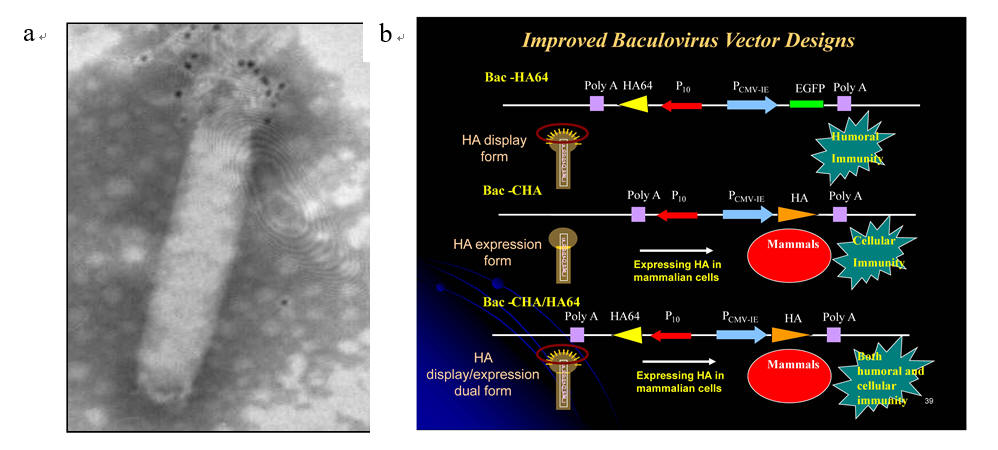
Fig. 4. Pseudotyped baculovirus as an avian influenza virus (AIV) vaccine. (a) The baculovirus (Bac-HA64) is engineered to display the hemagglutinin (HA) on the surface as indicated by the dots on the viral surface. (b) Development of baculovirus that can simultaneously display HA and express HA after entering mammalian cells (Bac-CHA/HA64). The new vector can induce both humoral and cellular immune responses, which is critical to the AIV vaccine efficacy.
Cancer Gene Therapy
We have engineered various hybrid baculovirus vectors to carry anti-cancer genes such as hEA (a fusion protein that inhibits new blood vessel formation), microRNA mimics/sponges and long noncoding RNA (lncRNA). Transduction of cancer cells by these vectors suppresses cancer cell proliferation, inhibits cancer cell migration/invasion, mitigates angiogenesis (new blood vessel formation), etc. Injection of the baculovirus vectors into tumors represses tumor growth, induces immune cell infiltration and inhibits blood vessel formation. These results remonstrates the potential of hybrid baculovirus vectors for cancer gene therapy.
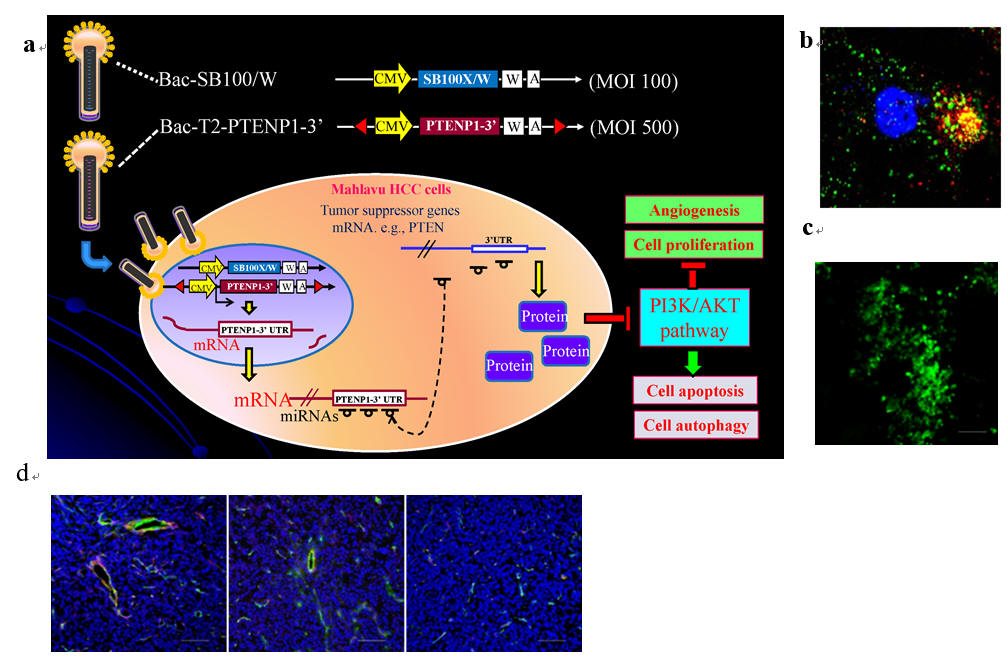
Fig. 5. Hybrid transposon-based baculoviral vector encoding PTENP1 lncRNA inhibits hepatocellular carcinoma (HCC) tumor growth in mice. Transduction of the aggressive HCC cells leads to sustained PTENP1 lncRNA expression, which rescues the tumor suppressor gene PTEN expression. Restored PTEN expression suppressed the downstream PI3K/AKT pathway, leading to enhanced cancer cell apoptosis and autophagy, while suppressing cancer cell proliferation and angiogenesis .
CRISPR-based Genome Editing, Regulation and Synthetic Biology
CRISPR-Cas9 is a powerful genome editing tool while CRISPR interference (CRISPRi) and CRISPR activation (CRISPRa) emerge as novel tools to modulate gene expression and genetic interaction. Our lab is currently combining CRISPR-Cas9, CRISPRi and CRISPRa to manipulate the metabolic networks in microorganisms (e.g. E. coli or cyanobacteria) so that the microorganisms can use carbohydrates derived from biomass (e.g. woodchips) or CO2 as the carbon source to produce new high value chemicals. We are also combining synthetic biology principles, CRISPRi and CRISPRa to regulate the differentiation fate so as to boost the stem cells differentiation and tissue regeneration.
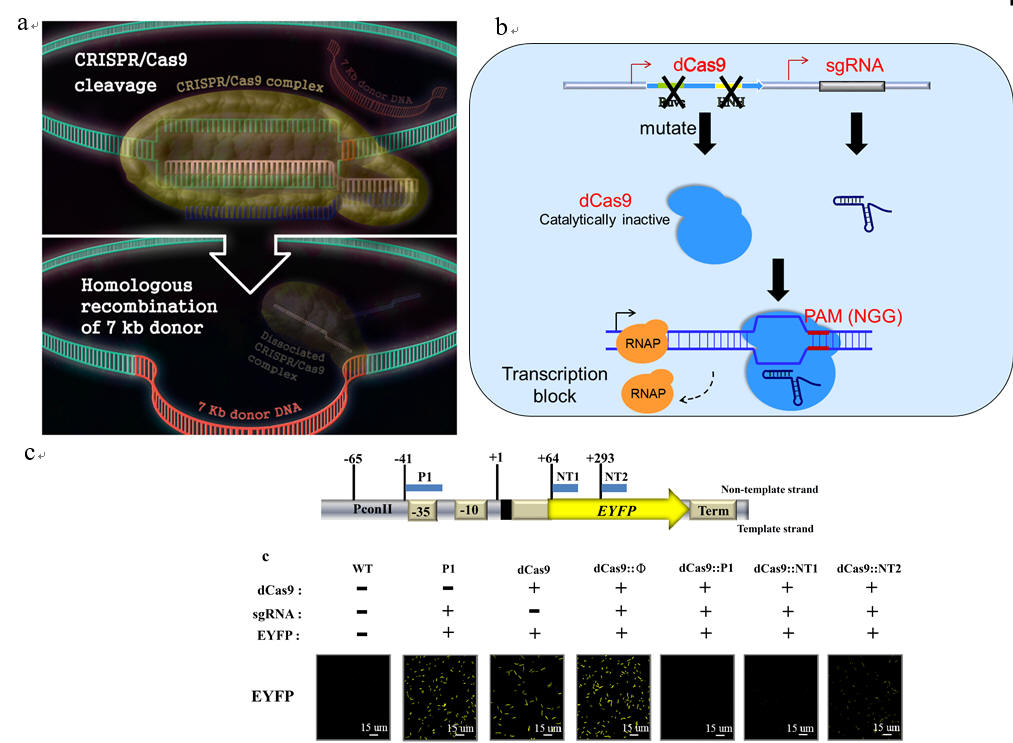
Fig. 6. CRISPR-Cas9-based genome engineering and manipulation. (a) CRISPR-Cas9-mediated DNA cleavage enables and facilitates the integration of a DNA as large as 7 kilobases (kb) into E. coli chromosome. (b) The catalytic domains of Cas9 are mutated to generate the inactive Cas9 (dCas9) lacking the endonuclease activity, laying the foundation of CRISPRi technology. By co-expression of dCas9 and a chimeric single guide RNA (sgRNA), the dCas9-sgRNA complex specifically binds to the target gene and acts as a roadblock to the elongating RNA polymerase (RNAP), hence aborting transcription initiation or elongation. (c) We designed sgRNA that targets the promoter (P1) or coding sequences (NT1 and NT2). Co-expression of dCas9 and sgRNA in cyanobacterium PCC 7942 confers effective and stable suppression of EYFP expression at efficiencies exceeding 99%, without impairing cell growth .